Anaphylaxis Shock Symptoms from Anaesthetic Caused by Cough Medicines
Anaesthetists in Australia are calling for many over-the counter lozenges and cough medicines to be restricted or banned from the Australian market because of the risk that they can trigger life-threatening allergic reactions during surgery.
Australian anaesthetists have asked Australian drug administration agencies to ban or reduce access to cough medicines containing the cough suppressant, pholcodine due to suspected links to Anaphylaxis (a severe allergic reaction) in patients given muscle relaxants during surgery.
Norwegian health authorities banned cough medicines containing pholcodine due to concerns about allergy risks. Since the ban was introduced, the rate of Anaphylaxis in Norway has decreased significantly.
About 53 common over the counter cough medicines in Australia contain this drug which acts as a cough suppressant.
People who take pholcodine, days or weeks before surgery, are thought to develop an allergy to muscle relaxant drugs that are used for many operations including gall bladder and appendix removals.
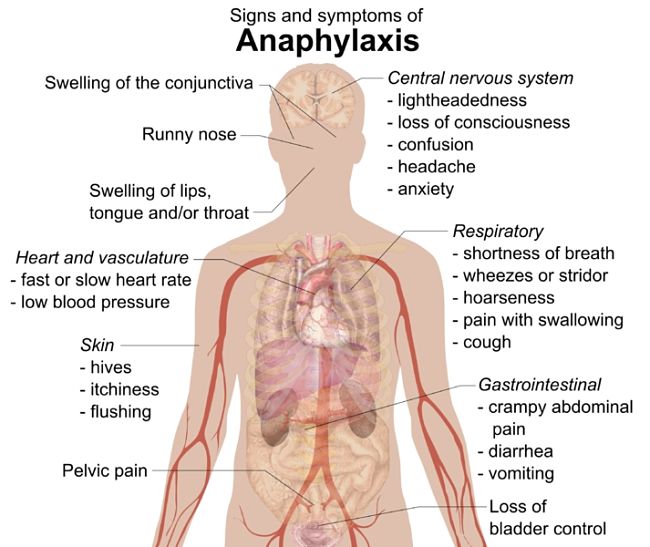
The reaction is similar to the severe allergic reactions that occur in some people to bee stings and eating peanuts. This article reviews the information on this risk.
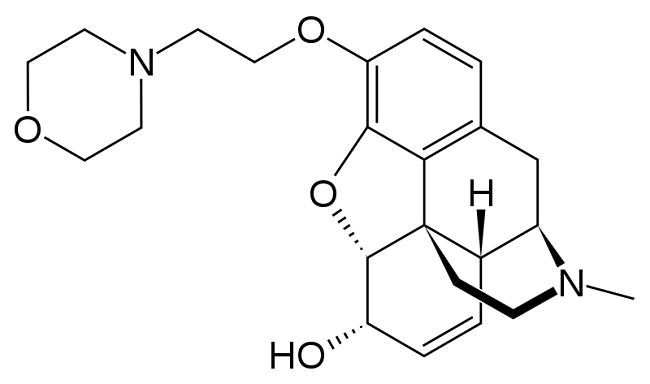
Pholcodine is a cough suppressant added to many over-the-counter cough medicines that acts on the central nervous system (CNS) depressing the
cough reflex. It also has a mild sedative effect, but no analgesic action.
A Norwegian study found that patients exposed to pholcodine
had a sharp rise in levels of IgE antibodies towards pholcodine,suxamethonium and morphine. The median proportional increase of
IgE was 19.0 times. The conclusion from the study was that pholcodine caused increases in IgE antibodies associated the development of allergies
towards Neuromuscular Blocking Agents (NMBA).
In Australia about one in every 10,000 surgery patients suffer
Anaphylaxis during surgery However the rate doubles to about one in every 6,000 when muscle relaxants were given during surgery.
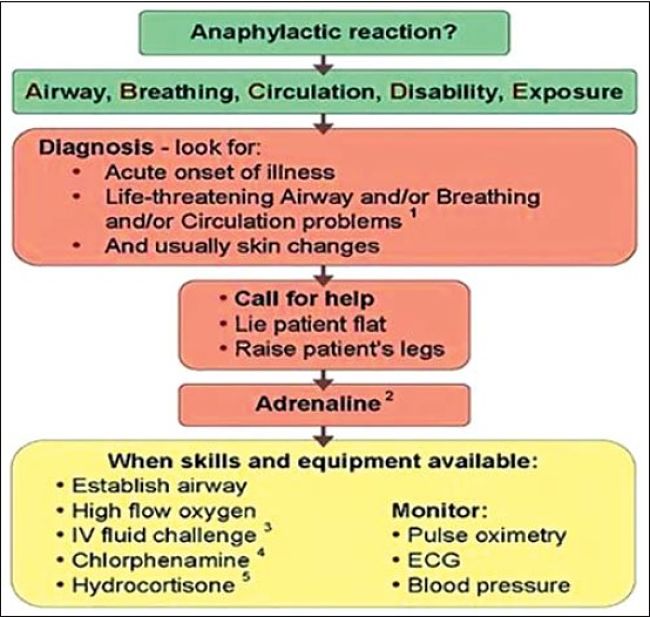
In some cases, the allergic reaction caused rapid cardiac arrest within minutes of administering the muscle relaxing drug.
While only about 1-2 % of patients die from the allergic reaction, it can cause severe brain damage and other problems.
The surgery may have to be abandoned and rescheduled. Such patients need special drug response testing before having surgery again.
There are 53 products containing pholcodine on Australia's register of therapeutic lists. Most are liquid cough medicines and cough lozenges,
that include including Duro Tuss, Demazin, Tixylix and Difflam products.
While scientists have not been able to prove conclusively
that pholcodine sensitivity triggers Anaphylaxis when patients are given muscle relaxants, but there is evidence of a link. In 2007, Norway banned
pholcodine products due to concerns about allergies. Since the ban, Anaphylaxis rates have declined significantly.
Other research has suggested that cough medicines were not very effective and advice has been issued that
many of them are unsuitable for infants under 2-3 years of age, for a variety of drugs included in the ingredients.
The anaesthetists argue that there were many alternative cough medicine products that do not contain pholcodine.
The risk of death and brain damage is too severe to be ignored despite its low incidence.
A review conducted by the European Medicines Agency the effectiveness
and safety and of pholcodine, in 2012, concluded the existing evidence of the risk of a link with Anaphylaxis during
surgery was weak and did not impose bans or restrictions for Europe.
So the jury is still out on this one.
Related Articles
=> Allergy Free Garden Design and Tips for Low Allergen Gardening
=> Best Natural Allergy Remedies - Identify, Avoid Seasonal Allergens
=> Cats Allergy Risk for Adults, Children - Ban Cats from Your Bedroom
=> Egg Allergy Symptoms - How to Avoid, Manage and Treat